
Herzinsuffizienz ist ein weltweites Gesundheitsproblem, von dem Millionen von Menschen betroffen sind. Was wäre, wenn es ein Gerät gäbe, das die Herzen der Patienten am Laufen halten und sogar ihre Lebensqualität verbessern könnte? In seinem Keynote-Vortrag auf der COMSOL Conference 2018 Boston sprach Freddy Hansen von Abbott Laboratories über eine Herzunterstützungspumpe, die genau das leisten könnte. Wenn Sie seinen Vortrag verpasst haben, können Sie sich hier die Video-Aufzeichnung ansehen und eine Zusammenfassung lesen.
Freddy Hansen spricht über die Verbesserung des Designs eines Linksventrikulären Unterstützungssystems für Patienten mit Herzinsuffizienz
Bessere Linksventrikuläre Unterstützungssysteme (LVAD)
„Die komplexeste Maschine, die jemals einem Menschen implantiert wurde“ – so beschrieb Freddy Hansen das HeartMate 3™ Left Ventricular Assist Device (LVAD), eine von Abbott Laboratories entwickelte Pumpe mit einem magnetisch schwebenden Rotor für Patienten mit Herzinsuffizienz. Bei Abbott arbeitet Hansens Team an der Verbesserung des LVAD-Designs, um den geschwächten Muskel in der linken Herzhälfte zu kompensieren und so potentiell tödliche Blutansammlungen im Herzen zu vermeiden. Während andere elektronische Implantate (wie beispielsweise Herzschrittmacher) nur einige Mikrowatt Strom verbrauchen, müssen LVADs den gesamten Blutstrom pumpen und verbrauchen daher eine beträchtliche Menge Strom, etwa im Bereich von 10 Watt. Weitere Herausforderungen bei der Entwicklung dieser Systeme sind, dass sie klein genug sein müssen, um in den Brustkorb eines Patienten zu passen, und dass sie bio- und hämokompatibel sein müssen. Um diese Probleme zu lösen, setzen Hansen und seine Gruppe Simulation ein.
Optimierung eines LVAD-Systems mit COMSOL Multiphysics®
Hansen erklärte, dass er COMSOL Multiphysics® verwendet, um alle Komponenten des LVAD-Systems zu simulieren. Zu diesen Komponenten gehören ein Elektromotor mit Magnetlagern, die einen Rotor im Inneren des Geräts in der Schwebe halten, sowie Sensoren und andere Elektronik, wie zum Beispiel die Steuerung des Elektromotors. Da jeder Teil dieses Systems lebenserhaltend ist, müssen die Ingenieure von Abbott sicherstellen, dass jede Komponente wie vorgesehen funktioniert. Das HeartMate 3™ LVAD ist der einzige Teil des Systems, der sich im menschlichen Körper befindet. Die Stromversorgung des Geräts erfolgt über Akkus und ein Kabel, das durch die Haut des Patienten geführt wird. Der Controller ist ein Patienten-Interface, das über Schaltflächen zur Überwachung der LVAD-Leistung verfügt (und auch Notfall-Akkus enthält).
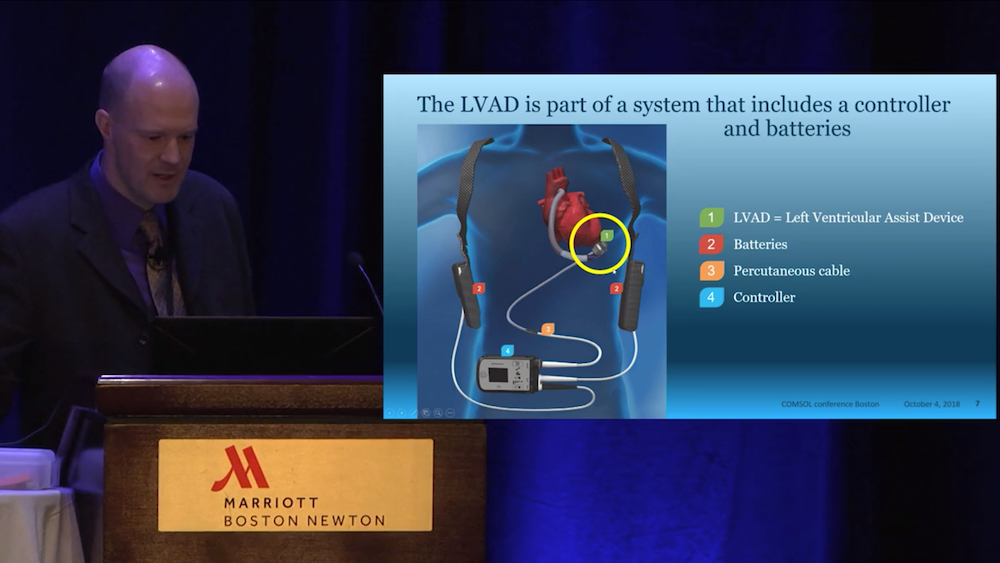
Aus dem Video: Ein LVAD-System. Das LVAD wird in den Brustkorb implantiert, während sich die übrigen Systemkomponenten außerhalb des Körpers befinden.
Hansen nannte mehrere Beispiele dafür, wie er und seine Gruppe verschiedene Modellierungstechniken einsetzen, um alle Komponenten und multiphysikalischen Interaktionen des LVAD-Systems genau zu analysieren. Bei der Entwicklung des HeartMate 3™ LVAD haben sie zum Beispiel ein Motormodell erstellt, das ein Magnetfeld im rotierenden Teil enthält, mit mehreren Spulen um den Kern, die den Rotor drehen und in der Schwebe halten. Sehen wir uns einige weitere Modellbeispiele an.
Modellierung des Wärmetransport im Controller und Implantat
Bei der Arbeit an dem LVAD-Design stellte Hansen eine wichtige Frage: Wie heiß wird die Haut in der Nähe des Controllers? Er wies darauf hin, dass es wichtig ist, die Temperatur des Systems auf der Haut zu bestimmen, denn der Patient kann das Gerät nicht einfach entfernen, wenn es zu heiß wird – es hält ihn oder sie am Leben! Um Antworten zu finden, entwarfen die Abbott-Forscher ein Biowärmemodell mit Schichten von Haut, Fett, Muskeln und Eingeweiden auf der einen Seite des Controllers und einer Kleidungsschicht auf der anderen Seite, um die thermischen Effekte zu simulieren. Das Modell verfügte sowohl über eine Fernkühlung des Blutes als auch über eine maßgeschneiderte konvektive Kühlung der Kleidungsschicht mit einem Wärmeübergangskoeffizienten.
Ein zweites Beispiel für ein thermisches Modell begann mit der Frage: Wie heiß wird das Gewebe neben dem Implantat? Um diese Frage zu beantworten, analysierte das Forschungsteam veröffentlichte Daten, indem es ein Experiment nachbaute, bei dem die thermische Konnektivität von lebendem Gewebe untersucht wurde. Als Nächstes simulierten sie das Einlegen des LVAD in ein Wasserbad und verglichen die Ergebnisse mit einem Vergleichstest. (Mehr darüber, wie die Temperatur der Akkus und anderen thermischen Komponenten überwacht wurde, erfahren Sie im Video).
Simulation der drahtlosen Stromübertragung
Das Abbott-Team untersuchte auch Möglichkeiten, das perkutane Kabel durch eine drahtlose Stromübertragung zu ersetzen – ein Ziel, das Hansen als den „heiligen Gral“ der Branche bezeichnete. Wenn dies gelingt, würde die drahtlose Stromübertragung das Infektionsrisiko an der Eintrittsstelle verringern. Das verbesserte Gerät würde nicht nur das Leben der Patienten retten, sondern auch ihre Lebensqualität erheblich verbessern, da die Patienten duschen und schwimmen könnten. Um diese Möglichkeit zu erforschen, entwickelte das Team ein Modell eines 3D-Magnetfelds und eines 0D-Stromkreises, um die Effizienz und den Energieverlust zu bewerten. Das Hinzufügen der elektrischen Schaltung war in diesem Fall wichtig, da der Stromkreis und die Magnetspulen stark wechselwirken.
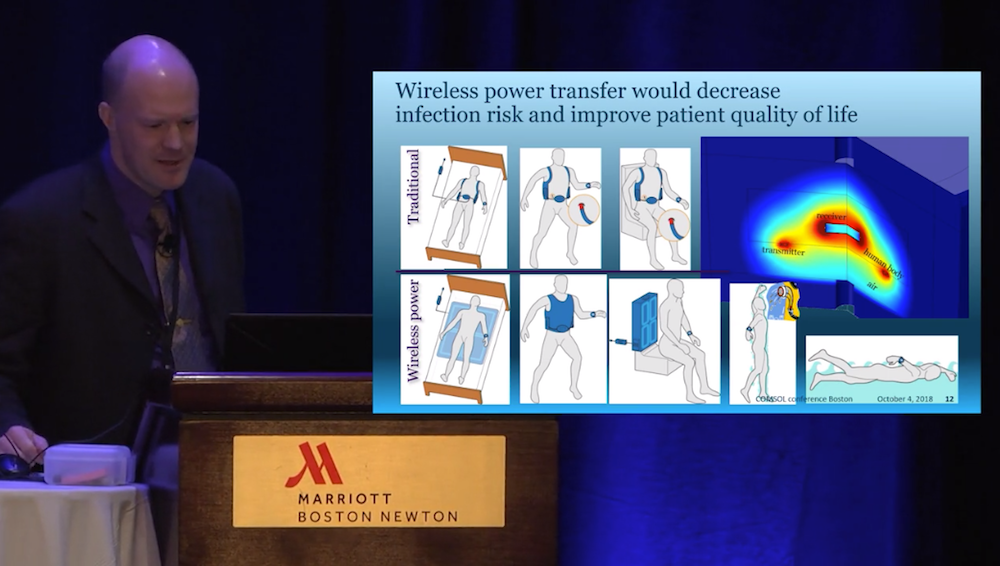
Aus dem Video: Das Modell für die drahtlose Stromübertragung.
Bewertung der Strömung von Blutzellen mit Simulationen
Hansen setzte seinen Vortrag fort, indem er erläuterte, wie die CFD-Modellierung dem Team half, den Weg des Blutflusses durch das Gerät zu verfolgen. Das Strömungsfeld durch das LVAD wurde als echtes multiphysikalisches Problem gelöst, da es interaktiv und zyklisch ist. Bei der Untersuchung der Blutzellenkonzentration muss die Scherbelastung berücksichtigt werden, da sich die Blutzellen von Orten mit höherer Scherbelastung wegbewegen. Diese Bewegung wirkt sich auf die Viskosität des Blutes aus, was wiederum das Strömungsfeld und die Scherspannung beeinflusst.
Bei der qualitativen Bewertung des Flusses für ihr LVAD-Design haben die Forscher darauf geachtet, dass kein Blut vom Rotor zurück in den Einlass fließt, da dies zu einer geringeren Effizienz und einem höheren Risiko von Blutschäden führt. Hansen sprach auch über die Verwendung der Partikelverfolgung zur Berechnung des „Waschens“, d.h. wie lange eine Blutzelle in der Herzpumpe bleibt. Sie fanden heraus, dass sie die Partikel nach der Entweichzeit sortieren und mithilfe der Simulation fortgeschrittene Blutschäden berechnen konnten.
Hansen schloss mit den Worten, dass er und das Abbott-Team für dieses Design mehr als 300 Modelle sowie Dutzende von Produkten und Prototypen verwendet haben. Die Simulation hat es ihnen ermöglicht, ein optimiertes LVAD-System zu schaffen und das HeartMate 3™ LVAD für den praktischen Gebrauch zu entwickeln.
Bisher haben die HeartMate LVADs mehr als 35.000 Leben von Patienten mit Herzinsuffizienz gerettet. Einige Patienten laufen Marathon und nehmen an vielen anderen Sportarten und Aktivitäten teil, aber das wirklich Beeindruckende an diesen Systemen ist: Sie halten Herzen am Schlagen und die Patienten am Leben.
Möchten Sie mehr über die Herzpumpensimulationen und das LVAD-Systemdesign von Abbott erfahren? Sehen Sie sich das Video am Anfang dieses Beitrags an.
HeartMate 3 ist eine Marke der Abbott Unternehmensgruppe.



Kommentare (0)